In 2013 Edward Snowden leaked thousands of classified American National Security Agency documents, sparking a global conversation about citizens’ rights to privacy on the Internet.
Week 1-5 in spring semester during junior
本栏目将从即日起将更新每周的生活、学习总结以及一些感悟,来刺激写博客的欲望,保持良好的记录、反思习惯。该博客于2014-01-13正式开通,起初的设想是为了锻炼雅思写作,尽量用英文写作。今后,在其他栏目尽量都用英文来练习。首先来总结一下博客的事情。
The simplest, the best
After building a blog with wordpress, I began to know more about domain, hosting, dns, blog platform. It was quite easy to establish your own website or blog with wordpress. However, I just found it too complex to use wordpress just for blog, personal blog in particular. With tons of developers contributing to themes and plugins, wordpress is so powerful that you can create almost everything you want. So, people tend to play with Ghost, tumblr, octpress……It seems everything returns to the origin, no php, no database, just static pages, just html, css, js. Then here comes the Medium in which you can immerse yourself in contents there. Simple search box, simple writing system, simple UI, no comments system, all you need to do are reading and writing.
As for nature science, “the simplest, the best” works as well. In order to explore the mystery of nature, to see things more clearly, scientists invent microscope, electron microscope, atomic force microscope…It becomes more and more difficult to handle these equipments. In addition, scientists determine Newton Law, Maxwell Electromagnetic theory, the Quantum theory. Undoubtedly, it becomes more and more tough for students to learn. Meanwhile the Unified Field Theory has been studied for ages. If the theory is complete one day, nearly every physical quantity will be unified together. No more atom, molecular, just some basic elements. Everything seems to return to the origin.
The rule goes for personal management too. Plan -> execution -> feedback. There are so many management software on Internet, such as Evernote, to do list, outlook……Finally, you’ll find just pen and pager are enough.
For education, MOOCs like Coursera, edX, Udacity emerge in large numbers. Many interesting parts are added to course materials. Similarly, paper books turns out a more efficient approach. Just like Lang Lang said “It’s impossible to practice piano with pleasure at the start. It takes long time and great efforts to play piano well. Then you can enjoy it”.
All in all, just follow the simplest way. Do not make things complicated.
Build python development environment in Notepad++
Introduction to python
Python is a programming language that lets you work more quickly and integrate your systems more effectively. You can learn to use Python and see almost immediate gains in productivity and lower maintenance costs.
Python runs on Windows, Linux/Unix, Mac OS X, and has been ported to the Java and .NET virtual machines.
Python is free to use, even for commercial products, because of its OSI-approved open source license. Official website for python is www.python.org. There are two versions of python, namely python 2.7.x and python 3.x. For more details, you can visit official website and download and install python according to your need. IDLE is the default IDE which contains editing, debug, shell…For a fresher, editing is enough. So I choose notepad++ as my editor.
Introduction to Notepad++
Notepad++ is a free (as in “free speech” and also as in “free beer”) source code editor and Notepad replacement that supports several languages. Running in the MS Windows environment, its use is governed by GPL License.
Python environment variable
- Computer -> System properties -> Advanced system settings -> Environment variables
- System variables -> Path -> Edit -> add ;C:Python27(or your installation directory of python) to Variable values -> OK
- Test if the setting works: Win+R -> input cmd -> input python
Notepad++ Settings
Run -> run -> input codes below -> save -> Name: Run Python(you can change as you prefer) -> OK
cmd /k python "$(FULL_CURRENT_PATH)" & PAUSE & EXIT
Test if it works
- New text -> renamed with “test.py” -> open “test.py” -> input
print "Hello World";
- New text -> renamed with “test.py” -> open “test.py” -> input
Enjoy editing python in Notepad++.
Thoughts on knowledge management
During high school in China, if you follow good teacher without hesitation, you’ll find it easy to do knowledge management. As for me, I was a guy who sometimes wanted to play trick and cut corners. However, most of the time, I’d like to listen to teachers’ words and I benefited a lot. In our high school, nearly everybody cared about mistakes and had several notebooks to collect mistakes to avoid them. At the very start, we wrote down mistakes from exams, exercises and right solution approach. Later on exams and exercises happened extremely frequently. It would waste plenty of time if we insisted writing all mistakes down. We began to cut the mistake parts from papers or books and paste them into our notebook. It was still a huge project, but worked. Now I guess, high school’s education focus on ability to solve problems, so we have exams and exercise books as vast as the open sea.
I’m a junior student at present with a year and a half left. High school education and college education are totally different. College education seems to concentrate on understanding of basic concepts and making connections between different concepts or subjects. In my eyes, it’s not worth that writing so many things due to courses in quantity and not wise to write everything in notebook like a carrier of textbook. There must be some more efficient ways to do knowledge management. For quite a long time, I enjoy writing few notes and doing more summaries. But, I seldom care about how to use my knowledge, namely output. With times go by, many useful things are buried and forgotten in my own notebook. In order to fix this up, I create this blog to share my understanding. I’m thinking over the best media to share. Words, mind maps, audio texts, videos. Words may be boring for readers sometimes. Mind maps are very intuitive and make collections between concepts clear. Audio texts, how to say, I haven’t tried yet, but I heard that one person made audio collections of his notes. Videos are quite good for learning new things or brushing up. There are plenty of tutorial videos in programming area, like HTML, JavaScript, PHP, Android, ios……few in natural science, such as chemistry, physics, biology. Perhaps it’s more difficult to describe natural science in videos, owing to some experiments you have to do in labs, not like coding in computers. Of course, there are many online education organizations including Cousera, edX, and other open courses which are pretty good in some degree.
Why not making my own video? I’m considering this. As a fan of dota, I usually watch wusheng2009’s video on Youku. Many retired professional dota player make comments on game with video. With more and more people interested in their video, they start to open clothes shop, snacks shop and the shop selling mouse, keyboard. Then, they put ads of their own shop into the video, and make a living by these. For me, I’m not going to make a living by these. I plan to output what I learn through video..maybe. Then I can get some comments from visitors.They point out my mistakes and I correct them, namely feedback. They point again and I correct again until everything is fine. Check and balance, check and balance again….like Gao Xiaosong said in Morning Talk II a talkshow on Youku.
Learning is a social and active process, even in the “hard” sciences!Learning is a social and active process, even in the “hard” sciences! Last updated: 2014.01.25
2013 Cambridge Summer School
Six months have passed since that fantastic summer experience. The memories are still fresh that I can remember many interesting things there.It was the first time for me to see the world outside China. I just couldn’t wait to feel the atmosphere there. To save money for our group, we transferred in Dubai and finally arrived in London. Then we would take a bus to Cambridge. Now I’d like to divide my experiences into several parts to describe in details.
Long journey by air
The two flights were both around 6 hours which were quite long especially in consideration of time difference. But the desire to get to Cambridge and the convenience in the plane just covered up the tiredness during the trip. I had to admit that the plane was so great, not only for its soft light adjusting to local time, but also the device containing movies, games, music, phone, and … in front of us.
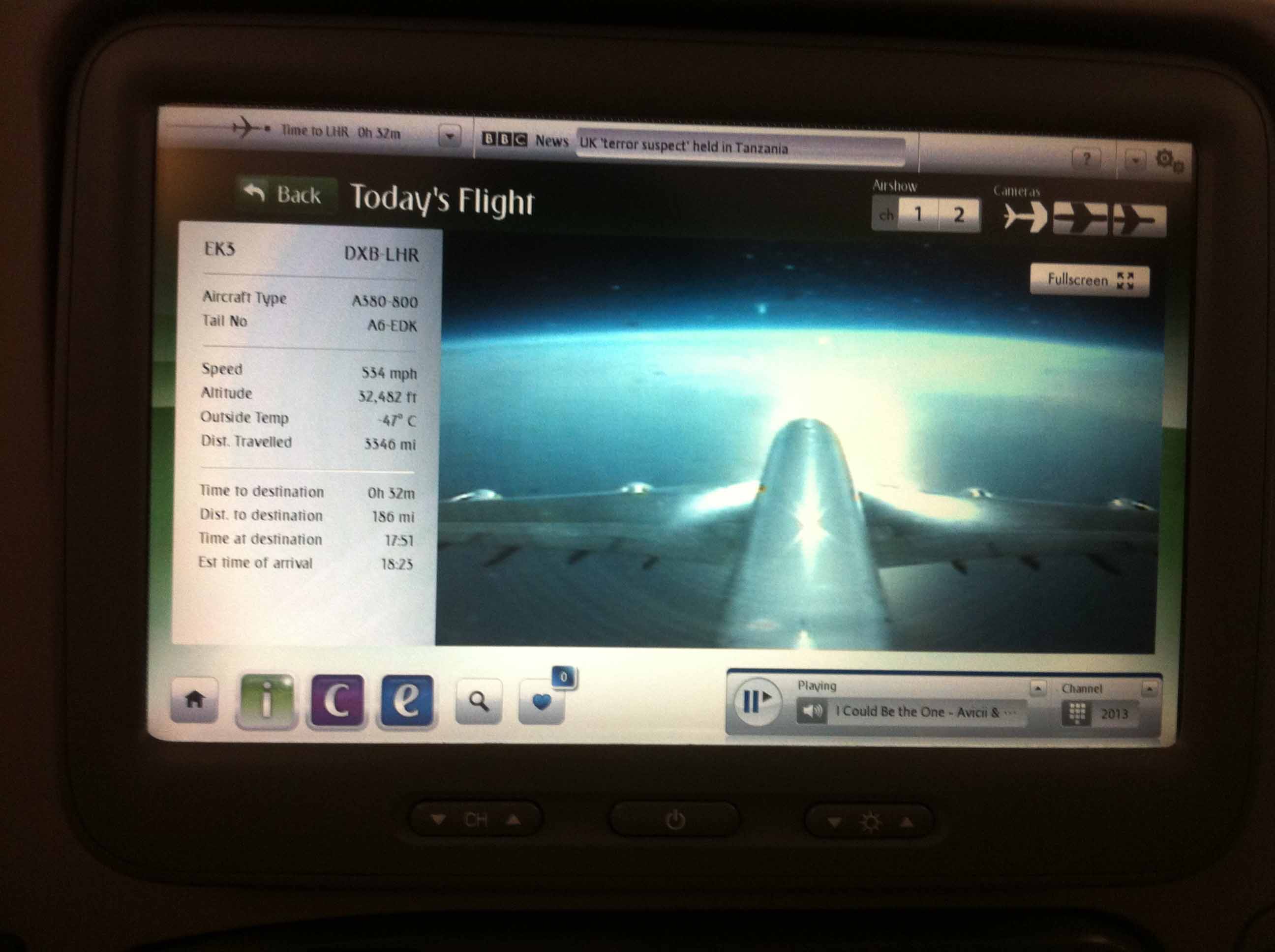
Flight camera
Watching movies, playing games, time went fast from Beijing to Dubai to London.
Beauty of Cambridge
I was drawn into the ancient buildings when I got there. In china, many old famous buildings were replaced by modern high buildings or destroyed. Just a few days ago, 2/3 ancient city area in Shangri-la were destroyed by fire, which caused a loss to over one hundred million RMB. How painful Ah. By contrast, Cambridge or other European cities did the preservation of cultural relics pretty well. In the World War II, people in Cambridge split the colorful glasses in the wall in small parts with unique numbers and transferred them from ground to basement. After war, people put them together according to the numbers. Owing to many great efforts like this, we can see the living history now.

Museum of Cambridge
The picture above showed a simple building in Cambridge, you could hardly see the building over six floors there. The road was narrow too, even one-way street somewhere. I couldn’t imagine these in Beijing.

Walking in the St. John’s College
One day we dropped into St. John’s College by the back door. There was a Shakespeare’s play in the garden. Although I was not able to understand what they were talking, I could feel the enthusiasm and concentration of actors. Plenty of trees, grass, old buildings, everything were great here.

Sitting on the grass
Sitting on the grass, enjoying the sunshine were both wonderful experiences in Cambridge.

XXX in line

Busy cam river
Statistical Molecular Thermodynamics
Basic information
- Source: Cousera
- University: University of Minnesota
- Link: https://class.coursera.org/thermodynamics-002
- Time: 2014.01.21~2014.03.25(nine weeks)
- Instructor: Dr. Christopher J. Cramer
- Books: Devoe, Howard. Thermodynamics and Chemistry.
- Focus: Chemical thermodynamics which is to say that we will delve into those thermodynamical details of greatest relevance to chemists,almost completely on thermodynamical properties and behaviors of gases.
- Syllabus: https://class.coursera.org/thermodynamics-002/wiki/syllabus
[toc]
How to learn something with Coursera
1.Be familiar with the course page
You should know where the course materials and homework are. Know the deadline of homework and how to submit your homework. Know where to discuss your problems with other people enrolled in the same course.
2.Manage your time well
It’s amazing to see some retired teacher involved in the course. Never too late to learn, right? However, I think most of people enrolled in the Coursera are students and young guys. They have their own work to do. So it’s very important to balance between the real world education and the online education or work. Luckily, in this course, Dr. Christopher J. Cramer put course materials two weeks ahead of schedule, which provides enough room to organize. There are about 8 videos a week.
3.Make full use of the video
There are various materials available, video, PDF of presentation, book. I used to read books to acquire knowledge, but prefer watching video after reading the guide of this course, and I benefit a lot. Because my native language is Chinese, not English, so it’s a little bit difficult for me to follow the course taught in English. But I believe the more I practice, the more I get, and language will not be a problem sooner or later. You may ask me how to make full use of the video.
Choose the speaking speed you can keep pace with Chris’s speed is proper for me, not hard to follow. Some new words come out sometimes, but they don’t influence me to understand. The best way to deal with the new words is skip them first, just put them down, and look them up in dictionaries after the video. In addition, you’d better read the homework before watching video, so you get key words for this week’s course and when you hear them in video you should slow down or turn on Closed Caption if you need.
Brush up from time to time For me, I can understand instructor’s words when watching videos. But the memories are not very strong. Sometimes I forget things totally because what I hear, what I say, what I see are all Chinese. So I need to write some notes available for me to review. This is why I’m writing this post. Key words and core concepts are fine, while too much details and explanation will make me look like the carrier of books. Making connections between different concepts and describing concepts with your own words are exactly the way to learn better.
Notebook
Week 1
Homework
Deadline: 23:59 PM on Friday, January 31, 2014 GMT
Key:
- V(R)=0 for the electronic energy of diatomic
- the wavelength of the emitted photon
- the bond dissociation energy,the vibrational frequency,the electronic energy
- unit conversion: cm^-1 —- KJ/mol
- excitation energy, state of the hydrogen atom
- the bond dissociation energy,the vibrational frequency,the electronic energy
- the energy diference between J=n and J=m
- Calculate the degeneracy
- Determine the number of various (translational, rotational, vibrational) degrees of freedom of N2
- Determine the number of various (translational, rotational, vibrational) degrees of freedom of C2H6
Notes
1.1 concepts
1.2 details
- the energetics of reactions are important for all chemical reactions
- asses change in enthalpy for a given direction
What’s so useful about thermodynamics? Thermodynamics has long been recognized as one of the most powerful and useful tools in chemistry and physics and all the physical sciences.
Goals for this course
- Learn how the Universe really works
- End up smarter than most politicians(see the video you’ll know the details) When the change in enthalpy is negative, the enthalpy is released and the heat is released. Then the reaction will move from the left to the right.
Quantum mechanics Quantum mechanics provides the foundational principles for all molecular processes. Molecular statistical mechanics builds upon that foundation, and itself serves as the basis for thermodynamics.In order to do thermodynamic, we need to know something results and key features from quantum mechanics.
Energy is quantized
- Only certain quantized energies are permitted for the electronic energy levels of atoms and molecules
- The quantization can be a powerful tool
1.3 new words
- rust: [n] a red or brown oxide coating on iron or steel caused by the action of oxygen and moisture
- alumina: Al2O3
- melt: [n&v] the process whereby heat changes something from a solid to a liquid
- fusion: [n] a transformation from a solid to a liquid
- emphasize: [v] to stress, single out as important
- spontaneous: [adj] happening or arising without apparent external cause
- wrap up: to complete or finish something
1.4 pronunciation
- kJ/mol : kilojoule per mole
- a * b: a times b
- j/mol·°C:
- E = hν: E equals h nu
- E = hc/λ: h times c divided by lambda gives the energy
- nu tilde
- 10^-18: 10 the minus 18th
- infinity
- h bar
Week 2
Homework
Deadline:23:59 PM on Friday, February 7, 2014 UTC
Key:
- application range of the hard-sphere potential
- B2v at a temperature above the Boyle temperature
- the compressibility factor
- calculate gas pressure given volume, mole, temperature, the compressibility factor
- the Boyle temperature
- B2v
- the correct expression for the Boyle temperature
Notes
2.1 concepts
- ideal gas equation of state
- extensive variable
- intensive variable
- R: universal gas constant
- triple point
- compressibility
- critical point
- van der Waals loops
- Maxwell equation loops
- Law of corresponding states
- reduced quantity
- Virial Equation of State
- the second virial coefficient
- molecular interaction
- hard sphere model
- square well model
2.2 details
Why gases?
T/°C = T/K - 273.15
- Low T and Low P: attractive forces
- High T and High P: repulsive forces
2.3 new words
2.4 pronunciation
: the limit as pressure goes to zero, pressure times molar volume divided by the universal gas constant
- 273.15K: 273 point 15 degrees Kelvin

